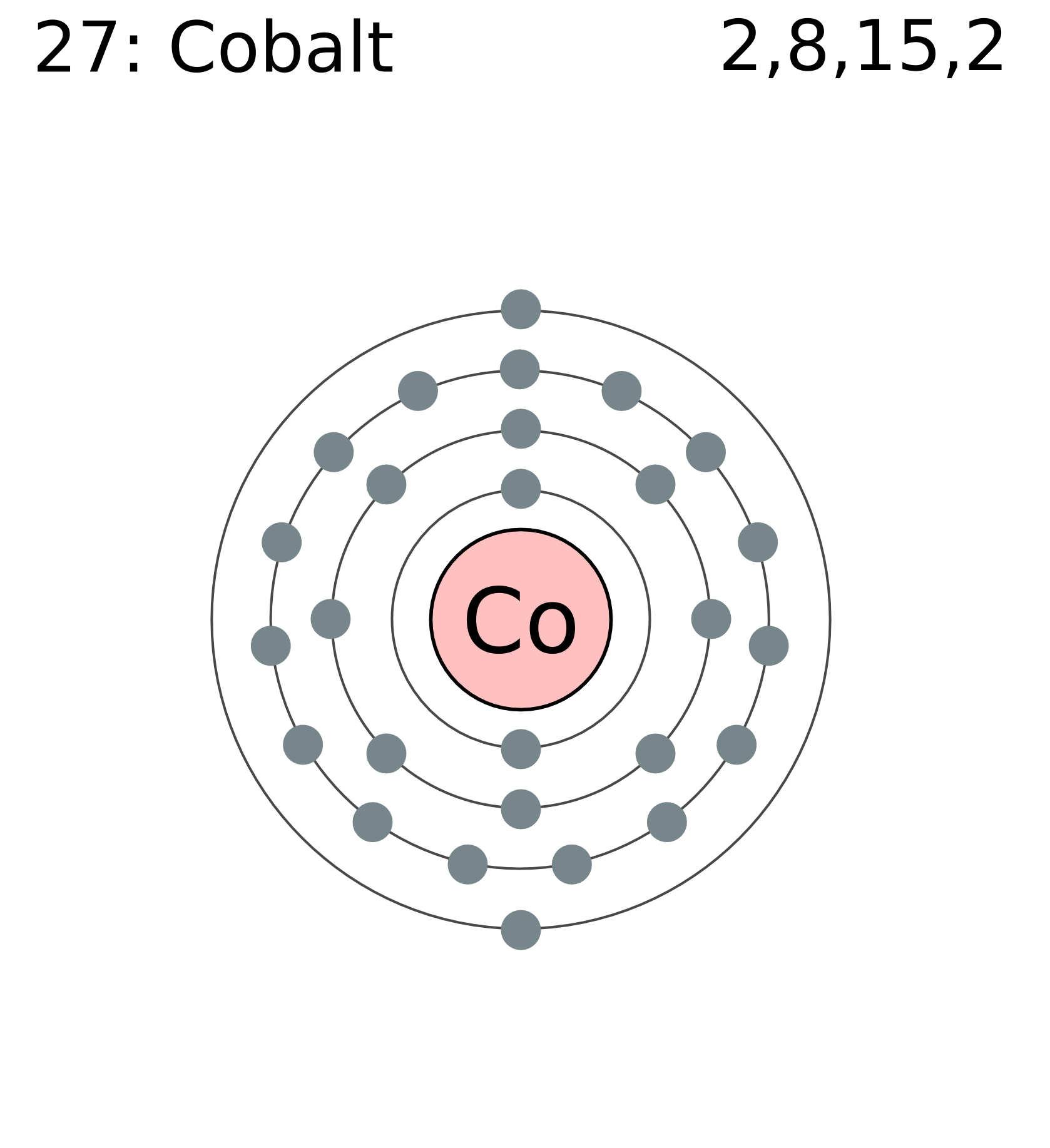 
Now it is possible to find the orbital notation of cobalt very easily through electron. The electron configuration of chromium can be expressed as Ar4s. The atomic number of cobalt is 27, which means it has 27 electrons. It is used in many places today, such as, magnets materials, paint pigments, glasses, and even cancer therapy. Cobalt was first discovered in 1735 by George Brandt in Stockholm Sweden. The atomic number of Cobalt is 27 with an atomic mass of 58.933195. Mårtensson, "Core-Level Binding Energies in Metals," J. (a) Explain why the relative atomic mass of cobalt is greater than the relative atomic. Cobalt (Co) lies with the transition metals on the periodic table. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The electronic configuration of cobalt is 1s2,2s2,2p6,3s2,3p6,3d7,4s2. Chromium (atomic symbol: Cr, atomic number: 24) is a Block D, Group 6, Period 4 element with an atomic weight of 51.9961. The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. A common oxidation state for simple compounds is +2 (cobalt (II)). Common oxidation states of cobalt include +2 and +3, although compounds with oxidation states ranging from 3 to +5 are also known. All values of electron binding energies are given in eV. Electron configuration of Cobalt is Ar 3d7 4s2. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. The electronic configuration of cobalt is 1 s 2, 2 s 2, 2 p 6, 3 s 2, 3 p 6, 3 d 7, 4 s 2. These effective nuclear charges, Z eff, are adapted from the following references: ChemTube3D contains interactive 3D chemistry animations and structures, with supporting information, for students studying some of the most important topics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
